The Evolving Medical Cannabis Market: A Closer Look at Recent Trends and Shifting Paradigms
The global medical cannabis market is undergoing a remarkable transformation. As regulatory frameworks improve and evolving public attitudes drive acceptance, we are entering an era where medical cannabis is positioned not only as an alternative therapy for chronic pain management but also as a potential game-changer in the treatment of neurological and mental health conditions. In this editorial, we will dig into the latest market data, regulatory moves, and product innovations that are reshaping the field, while also addressing the tricky parts researchers and clinicians face along the way.
Growing Acceptance and the Power of Scientific Advancements
Over the past several years, scientific research has played a key role in pushing cannabis into mainstream medical research. Breakthrough studies have begun to highlight the potential of cannabis-based medicines for conditions like chronic pain, epilepsy, and even complications arising from chemotherapy. In parallel, clinical trials are providing valuable insights into both the benefits and the confusing bits associated with cannabis treatment.
Recent research initiatives include double-blind randomized controlled trials, such as those recently launched by University College London and Great Ormond Street Hospital. These studies aim to evaluate the combined use of tetrahydrocannabinol (THC) and cannabidiol (CBD) in treating epilepsy across various age groups. The trial’s outcomes may simplify the tangled issues faced by regulatory bodies in approving cannabis-derived treatments for broader patient groups.
Researchers are now poking around the hidden complexities of how cannabis compounds interact with the human body. With careful observation of fine points like pharmacokinetics and therapeutic windows, scientists are slowly building a better case for cannabis use in both mental and neurological health management. As public opinion shifts in favor of alternative therapies, the tie between scientific breakthroughs and regulatory reform grows ever stronger.
Chronic Pain and Neurological Health: Are We on the Brink of a Revolution?
One of the aspects generating the most buzz in the medical cannabis market is its application in chronic pain management and neurological health. Patients suffering from neuropathic pain, arthritis, and similar conditions are beginning to have more options, especially when traditional treatments prove overwhelming or ineffective.
Medical cannabis offers an intriguing alternative to opioids, providing patients with a less addictive option to manage chronic pain. Moreover, the therapeutic role of cannabis in addressing neurological issues such as Parkinson’s disease symptoms, multiple sclerosis, and epileptic seizures is being brought to the forefront of medical discussion. The opinion among many healthcare professionals today is that we have reached a tipping point where medicinal cannabis may well become a cornerstone of pain management therapies.
It is important to acknowledge that these developments do not come without challenges. Critics highlight the off-putting gaps in clinical knowledge and dosing guidelines, which can lead to a nerve-racking prescribing environment for clinicians. Yet, with further research and more controlled trials, these confusing bits are expected to be clarified, allowing both patients and doctors to confidently figure a path towards effective treatment plans.
Regulatory Shifts: New Policies and a Global Perspective
Government initiatives and regulatory reforms are fueling growth in the medical cannabis market. The legal framework in many regions is evolving, with various countries and states updating their policies to allow easier access for patients and to streamline the research and development process. For instance, recent changes in North America have led to an increased number of states permitting the prescription of medical cannabis.
In a significant move, the Therapeutic Goods Administration (TGA) in Australia has introduced a special access scheme, enabling medical practitioners to prescribe cannabis-based medicines more efficiently. Similar steps are seen in European nations, where regulatory bodies are formulating strict guidelines regarding the production, development, and distribution of cannabis therapies.
A noteworthy example of proactive regulation can be seen in Ukraine. In August 2024, the country officially launched a medical cannabis program, followed by further policy updates in September 2024, which signaled a robust commitment to improving patient access while ensuring strict oversight. Such moves demonstrate that governments worldwide are increasingly willing to work through the tangled issues associated with cannabis regulation to embrace its potential medical benefits.
Product Innovations: Dissolvable Forms, Oils, and Versatile Delivery Options
There is a wide array of products available in the medical cannabis market, each with its own set of subtle parts and benefits. In 2024, dissolvable forms and powders dominated the market due to their improved absorption rates and enhanced bioavailability. These formats offer flexibility in dosing and ease of consumption, making them particularly attractive for patients looking for a discreet, user-friendly option.
At the same time, oil-based products have emerged as a strong contender, showing the fastest growth in the predicted timeframe. Oils have diverse industrial applications, finding roles not just in pharmaceuticals, but also in cosmetics, agriculture, and even as alternative energy sources in some chemical processes. Here’s a quick comparison of the main product types:
| Product Form | Key Advantages | Main Applications |
|---|---|---|
| Dissolvable/Powder | Improved absorption, dosage flexibility | Medical consumption, easy administration |
| Oils | Versatile, strong growth potential, industrial applications | Pharmaceuticals, cosmetics, agriculture |
| Solids, Ointments & Creams | Topical application, localized effect | Skin treatments, localized pain relief |
In terms of derivatives, cannabidiol (CBD) has carved out a dominant position because of its promising anti-inflammatory and neuroprotective effects. Meanwhile, tetrahydrocannabinol (THC) is predicted to grow quickly, especially as regulatory bodies, such as the U.S. Food and Drug Administration, increasingly approve its use in treating chemotherapy-induced nausea and other conditions.
These product innovations underline how companies are continuously adapting to meet the diverse needs of patients while simultaneously addressing the subtle differences in product efficacy and patient preference. The ability to choose from a broad array of therapies is turning out to be a must-have feature in today’s competitive medical cannabis landscape.
Delivery Methods: From Inhalation to Oral Medications
The route by which a medication is administered can make a significant difference in its effectiveness and patient compliance. In the medical cannabis market, two routes stand out: inhalation and oral administration.
The inhalation route, thanks to its rapid onset of action and targeted delivery, has consistently dominated market share. It allows patients to achieve quick therapeutic effects, a factor that is particularly important in acute pain management. However, inhalation may not suit everyone, especially those with respiratory conditions.
On the other hand, oral forms of cannabis are experiencing rapid growth owing to their cost-effectiveness and reliability in achieving sustained release of active compounds. Oral medications eliminate the need for specialized equipment, making them an accessible option for patients who prefer a more straightforward approach to medication management. The different delivery options provide a range of solutions that can be tailored to individual patient needs, ensuring that both rapid onset and long-lasting relief are achievable goals.
Understanding the Challenges: Tricky Parts and Tangled Issues in Clinical Research
No discussion about the medical cannabis market would be complete without addressing the many tricky parts that researchers face in this evolving field. Despite the promise that cannabis-based treatments hold, public studies have found several confusing bits that inhibit progress.
- Funding Shortages: Many publicly funded trials are constantly battling limited budgets, which can delay the introduction of critical findings.
- Regulatory Hurdles: The emerging nature of cannabis as medicine means that researchers must contend with stringent regulatory requirements that vary across regions, making multi-national studies particularly challenging.
- Logistical Challenges: Coordinating between different institutions and managing the supply of regulated products can be an intimidating task.
These off-putting obstacles, while real, are progressively being worked through by collaborative efforts amongst governments, private entities, and research institutions. Over time, it is expected that these nerve-racking hurdles will diminish as protocols are standardized and funding streams become more reliable.
Distribution Channels and Patient Access: Opening Doors Through Online and Pharmacy Networks
Access to medicinal cannabis is regulated not only by legal frameworks but also by the channels through which it is distributed. Hospital pharmacies have historically been at the forefront in providing regulated cannabis products. Their involvement has been crucial for patient safety and cost-effective treatment options, bolstered by the expertise of trained pharmacists who can manage specific dosing requirements.
Online pharmacies are emerging as a viable option, particularly for patients in remote areas or those who prefer the convenience of digital access. The growing popularity of telemedicine, combined with robust patient portals and dosage tracking tools, is making it easier than ever for patients to get the medicines they need. These online platforms are proving to be a valuable complement to traditional distribution channels, ensuring that patient care remains consistent regardless of patient location.
Here’s a summary of the key distribution channels in the medical cannabis market:
- Hospital Pharmacies: Ensure expert consultation and regulated access.
- Online Pharmacies: Expand reach, particularly in underserved or rural populations.
- Retail Pharmacies: Offer flexibility and ease of access to a wide range of patients.
As more regions adopt these distribution models, we can expect increased patient engagement and enhanced adherence to treatment regimes, thereby unlocking even greater potential for medicinal cannabis in everyday medical practice.
Geographical Outlook: Insight into Regional Trends and Their Impact on the Market
One cannot fully understand the medical cannabis market without taking a closer look at the regional dynamics that are at play. The market performance is not uniform across the globe; instead, it varies considerably from one region to another due to differences in regulation, research funding, and cultural attitudes toward cannabis-based medicine.
North America: Leading the Charge in Regulation and Adoption
North America remains a critical market for medical cannabis, primarily because of its progressive regulatory environment. Recent legislative changes in the United States have paved the way for the medical use of cannabis in several states and territories. For example, state-level initiatives such as the recent state medical cannabis laws introduced by the National Conference of State Legislatures have boosted market legitimacy. In the U.S., advances such as proposals to reschedule cannabis from a controlled substance category further underline the growing acceptance of this treatment option.
Canada continues to play an important role as well, with the Government of Canada and Health Canada maintaining stringent data collection and regulatory standards via the Cannabis Act and related regulations. Such systems are designed to ensure that only safe and effectively monitored cannabis products are available to patients, reinforcing public trust in the medical cannabis system.
Asia Pacific: Rapid Growth Amidst Government Support and Legalization
The Asia Pacific region is emerging as one of the fastest growing in the global medical cannabis market. With countries beginning to recognize the therapeutic merits of cannabis, government endorsements and supportive policies have spurred rapid growth. India, for instance, is witnessing a surge in cannabis HealthTech companies, with the Central Drugs Standard Control Organization introducing new processes to facilitate both medical and non-medical uses of cannabis products.
Thailand, through unique partnerships in January 2025 with prominent pharmaceutical companies, is positioning itself as a regional hub for high-quality medicinal cannabis products. Meanwhile, fast-growing economies such as Singapore and Japan are also exploring regulated frameworks that could make medical cannabis more accessible to patients in the near future.
Europe and Latin America: A Diverse Regulatory Landscape
Across Europe, countries face a mixed bag of regulatory standards that affect how cannabis products are produced, developed, and distributed. Nations such as the United Kingdom, France, and Germany are in the midst of revising guidelines to carefully balance patient access with safety. The focus is on ensuring that innovations in cannabis-based treatments have a clear path forward, amidst rigorous national controls.
In Latin America, countries like Brazil, Mexico, and Argentina are gradually opening up their regulatory frameworks. Although these markets still have some tense issues to resolve, the overall trend is toward increased research funding and gradual legalization, which suggests promising future prospects for medicinal cannabis in these regions.
Strategic Moves by Key Industry Players: Steering Through a Competitive Landscape
Major players in the medical cannabis market are actively pursuing strategies designed to capture larger shares in this expanding market. Companies such as Canopy Growth Corporation, Aurora Cannabis, Tilray Brands, and Cronos Group continue to innovate in both product development and market penetration strategies. By targeting high-demand products like high-THC flower, vapes, edibles, and pre-rolls, these corporations are ensuring that they remain at the cutting edge of market evolution.
Recent revenue reports, such as those from Green Thumb Industries, attest to the rapid commercialization of these products, even as they adhere to strict financial reporting norms. These strategic moves are not without their challenges, as companies need to manage your way through a variety of tricky parts, including regulatory changes and competitive pressures. Nevertheless, their continued investment in research and development is a testament to the underlying belief in the long-term potential of medical cannabis.
A quick overview of some key industry participants and their contributions:
- Canopy Growth Corporation: Focused on high-demand medical products and ongoing product innovations.
- Aurora Cannabis Inc.: Building a robust portfolio aimed at various diagnostic and therapeutic applications.
- Tilray Brands: Emphasizing both research and strategic global expansion initiatives.
- Cronos Group Inc.: Investing in technologies to streamline production and expand product offerings.
This competitive landscape illustrates how industry leaders are more than ever committed to providing effective, accessible, and safe cannabis-based therapies, even as they work through tangled issues in clinical research and regulatory harmonization.
Challenges Ahead: Funding Struggles, Regulatory Bottlenecks, and Clinical Considerations
While optimism is high, it is important to take a balanced view by acknowledging the nerve-racking obstacles that remain. For instance, funding shortages continue to plague many clinical research projects, limiting our understanding of the full potential of cannabis-based treatments. Moreover, logistical challenges often slow down critical research when compared to other established medical therapies.
Another significant challenge is the need for standardized clinical protocols covering everything from dosing to long-term safety. These confusing bits are especially problematic in areas where the regulatory environment is still evolving. Without a uniform set of guidelines, clinicians often find it difficult to figure a path that provides both efficacy and safety in treating a wide range of conditions, from pain management to neurological disorders.
Nonetheless, patients and healthcare providers are gradually working through these issues. On the bright side, as governments and research bodies increasingly collaborate, there is hope that these complicated pieces will soon become more manageable. Initiatives across major markets have invested in initiatives designed to streamline funding, reduce regulatory ambiguity, and provide robust data to support the safe use of medicinal cannabis.
Future Prospects: The Critical Role of Innovation, Data, and Policy Reform
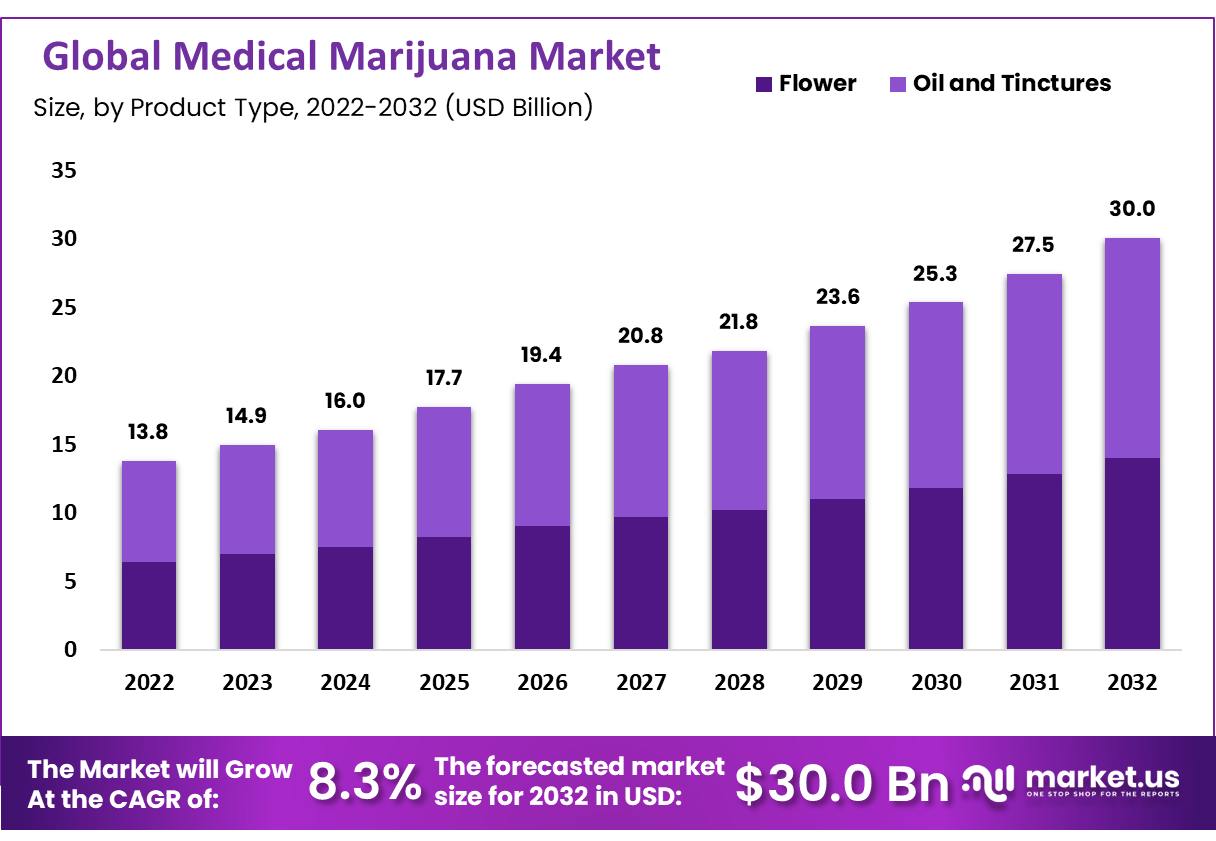
The future of the medical cannabis market appears layered with both promise and challenges. As market size projections predict substantial growth – from an estimated USD 14.34 billion in 2024 to upward of USD 52.23 billion by 2034 – the key to success will lie in a balanced approach that includes effective policy reform, continued innovation, and deep clinical research.
Central to this development is a comprehensive data-driven strategy. By combining insights from market research reports with firsthand clinical evidence, stakeholders can take a closer look at the subtle details that determine treatment efficacy and patient safety. Data collection systems are being continuously refined by government agencies such as Health Canada, whose Cannabis Tracking System Order and related measures ensure that product quality and regulatory compliance remain uncompromised.
In addition to data, strong legal frameworks are necessary to shore up consumer confidence. Government measures like the TGA special access scheme in Australia and recent legislative moves in the United States underscore the critical role that policy plays in legitimizing and fostering growth. These policies are not without their challenges, but by working through the tangled issues, governments are showing that they are committed to providing a safe and supportive environment for medical cannabis innovations.
In summary, the future of the medical cannabis market will depend on the combined efforts of researchers, industry leaders, legislators, and patient advocacy groups. By creating a collaborative ecosystem, the sector stands to benefit from enhanced clinical studies, streamlined research funding, and ultimately, improved patient outcomes, with each stakeholder contributing to a more robust and active market landscape.
Turning Challenges into Opportunities: Insights for Stakeholders and Policymakers
For both seasoned investors and new players looking to get their footing in the medical cannabis market, the current environment offers a wealth of opportunities. However, as with any rapidly evolving sector, prospective stakeholders must be prepared to work through several nerve-racking challenges.
Here are some key recommendations for those interested in leveraging the opportunities presented by the market:
- Invest in Research and Development: With many clinical trials in progress, opportunities abound for innovation in product delivery systems, such as oil-based formulations and dissolvable products. Investing in research can help reduce the intimidating gaps in current knowledge.
- Embrace Regulatory Changes: Stay informed of the latest legal developments. Engaging with policymakers and participating in industry associations can help businesses get ahead of regulatory changes and figure a path through the evolving landscape.
- Enhance Patient Access: Leveraging both traditional pharmacy channels and online platforms can ensure better reach, particularly for remote patients. This multi-channel approach also reduces the risk of supply disruptions and offers a broader range of treatment options.
- Prioritize Data-Driven Strategies: Use market research and clinical data to guide product development and marketing strategies, ensuring that the therapeutic benefits of cannabis are communicated effectively to both healthcare providers and patients.
For policymakers, the message is clear: continued support through funding, easing of some regulatory bottlenecks, and a collaborative approach to safety and efficacy guidelines will be super important in ensuring that the potential of medical cannabis is fully realized.
Public Perception and Societal Shifts: Changing Attitudes Toward Cannabis-Based Treatments
The stories surrounding medical cannabis have long been surrounded by stigma and misunderstanding. However, the gradual shift in public perception over the past decade has played a critical role in transforming the market. As patients, physicians, and researchers become more informed about the therapeutic merits of cannabis, we are witnessing a shift away from past misconceptions.
Social media, patient testimonials, and increased media coverage are all contributing to demystifying cannabis as a medicine. This smoother flow of information helps reduce the intimidating image of clinical trials and complicated regulatory hurdles, thereby making it easier for the general public to support and adopt cannabis-based treatments. With time, these gradual transformations are expected to further catalyze an environment where cannabis becomes a normal part of modern healthcare.
Looking Ahead: The Road to Mainstream Adoption
Beyond the immediate benefits for chronic pain and neurological conditions, the long-term promise of medical cannabis lies in its potential to revolutionize a broad range of therapeutic areas. The ongoing research aims to extend its applications to mental health management, inflammatory conditions, and even certain dermatological ailments. For instance, studies suggest that cannabis may have anti-inflammatory and antioxidant properties that could help manage conditions such as arthritis and psoriasis.
As more evidence becomes available, clinicians will be better equipped to fine-tune treatment protocols that skip through the confusing bits of trial and error. This will not only lead to more personalized treatment plans but also enable better monitoring of patient outcomes. In addition, successful research outcomes and regulatory approvals will pave the way for a more widespread acceptance of cannabis-based therapies, reducing the overall fear and hesitation among both patients and medical practitioners.
The future may well see medical cannabis transition from a niche treatment option to a mainstream therapeutic tool, capable of addressing a variety of conditions with appropriate formulations and tailored dosing regimens. This evolution is contingent on sustained efforts from all stakeholders to work through the nerve-racking but potentially rewarding challenges that lie ahead.
Conclusion: A Promising Future Despite the Twists and Turns
In conclusion, the global medical cannabis market stands at the cusp of a profound transformation. With robust scientific evidence, increasing regulatory acceptance, and a growing portfolio of product innovations, the landscape is rapidly shifting towards a future where cannabis-based treatments will play a key role in modern medicine.
Although the path is laden with challenging parts, funding shortages, and tangled regulatory issues, stakeholders across the board—whether policymakers, clinicians, or investors—are committed to working through these hurdles. Much like navigating a complex maze, the journey requires patience, precision, and a willingness to learn from every twist and turn.
As research breakthroughs transform our understanding of both the benefits and the subtle details of cannabis-based therapies, it is clear that the future of the market is not only bright but also filled with promise. By taking a balanced approach that incorporates data-driven insights, innovative product development, and supportive legal frameworks, the medical cannabis sector can redefine patient care and open new horizons in therapeutic treatments.
For those who are prepared to embrace and invest in this dynamic field, the opportunities are as extensive as they are exciting. The key lies in continuing to figure a path through the tricky parts while fostering close collaboration between research institutions, industry experts, and regulatory authorities. In doing so, we can ensure that the full potential of medical cannabis is realized, benefiting countless patients around the globe.
In the end, this period of transformation, while occasionally off-putting and nerve-racking, invites us to look beyond the current limits and imagine a future where effective, accessible, and safe cannabis-based medicines are integral to our healthcare systems. As the industry matures, only time will tell just how far this revolution can go—and one thing is certain: the journey is only just beginning.
Originally Post From https://www.precedenceresearch.com/press-release/medical-cannabis-market
Read more about this topic at
Cannabis market data
Data on cannabis for medical purposes

Social Plugin